The most common monosaccharides have three to eight carbon atoms. Wohl degradation of C forms D which is oxidized by nitric acid to an optically active aldaric acid.

Aldopentose An Overview Sciencedirect Topics
Aldohexoses A and B are formed from aldopentose C via a KilianiFischer synthesis.

. Structure and Nomenclature The general formula CnH2nOn with one of the carbons being the carbonyl group of either an aldehyde or a ketone. The suffix-ose indicates that a molecule is a carbohydrate and the prefixes tri- tetr- pent- and so forth indicate the number of carbon atoms in the chain. Nitric acid oxidizes A to an optically active aldaric acid B to an optically inactive aldaric acid and C to an optically active aldaric acid.

Aldopentose An Overview Sciencedirect Topics
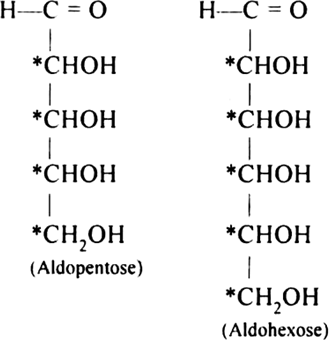
Draw Open Chain Structure Of Aidopentose And Aldohexose How Many Asymmetric Carbons Are Present In Each From Chemistry Biomolecules Class 12 Jharkhand Board

Aldopentose An Overview Sciencedirect Topics

Solved Consider The Structures Of The D Aldopentoses A Identify The Aldope Solutioninn
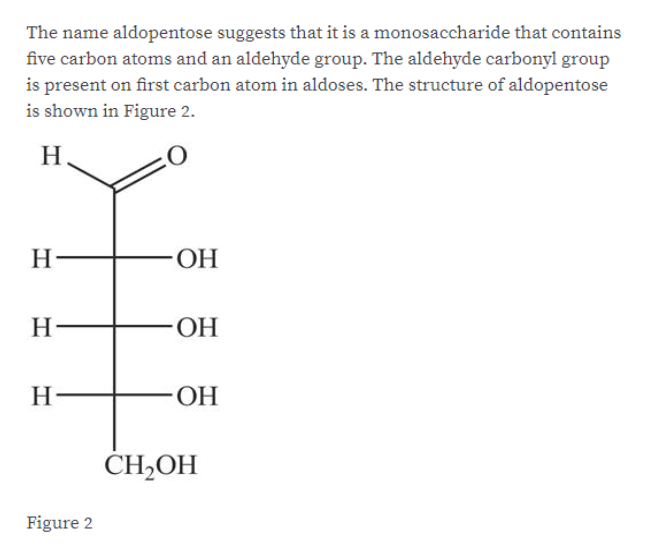
Answered Draw The Structure Of A A Bartleby
Solved Draw The Structure Of An Aldopentose Draw The Chegg Com

Solved 2a Draw The Structures Of Any D Aldopentose And Chegg Com
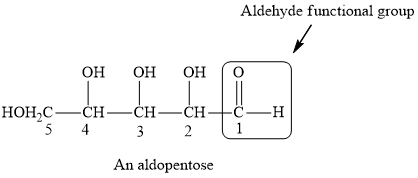
Solved Chapter 21 Problem 2p Solution Masteringchemistry Standalone Access Card For Fundamentals Of General Organic And Biological Chemistry 7th Edition Chegg Com
0 comments
Post a Comment